Expanded Umbilical Cord Blood Could Support Bone Marrow Transplants
Researchers at the Duke Cancer Institute recently reported results from a clinical trial of ex vivo expanded umbilical cord blood (UCB). The researchers tested expanded UCB use on high-risk patients with a history of blood cancers. Study patients did not have access to a matched, related or unrelated adult bone marrow donor. Those participating in the study ranged in age from 12 to 65 years.
 UCB is a rich source of stem cells, but the limited quantity of stem cells significantly reduces the utility of UCB stem cell transplants in adults. Bone marrow transplants are more common treatments for adults, but they require a closely matched donor. To increase the number of stem cells available to adult recipients, doctors may transplant multiple units of cord blood. And as seen in this trial, doctors may also be able to increase stem cell counts by expanding stem cells in a lab prior to transplantation.
UCB is a rich source of stem cells, but the limited quantity of stem cells significantly reduces the utility of UCB stem cell transplants in adults. Bone marrow transplants are more common treatments for adults, but they require a closely matched donor. To increase the number of stem cells available to adult recipients, doctors may transplant multiple units of cord blood. And as seen in this trial, doctors may also be able to increase stem cell counts by expanding stem cells in a lab prior to transplantation.
The Duke research sought to determine the safety of expanded stem cell transplants into adult patients. In this experiment, researchers cultured UCB stem cells for three weeks prior to transplant. On average, the culturing process expanded the number of available stem cells by 3300%. Researchers then transplanted a single dose of the expanded stem cell product, NiCord, into 36 patients.
During the trial, researchers compared the study patients to a prior study, whose patients received unexpanded cord blood. The patients in the Duke study were carefully matched to those in the first study.
Positive results from expanded UCB stem cell transplants
Nearly all (94%) of patients in the Duke study showed signs of successful engraftment six weeks after the UCB transplant. Significantly, the engraftment occurred faster in the Duke study patients than in patients who received unexpanded cord blood. This is important because the speed of engraftment appears to reduce the number of post-transplant complications. In addition, study patients achieved platelet recovery nearly twice as fast as those who received unexpanded UCB transplants. This decreased recovery time should reduce the amount of time patients stay in the hospital, one of the largest costs involved with cord blood transplants. The reduction in recovery time could quite possibly make this method the most cost-effective option for hematopoietic stem cell transplantation.
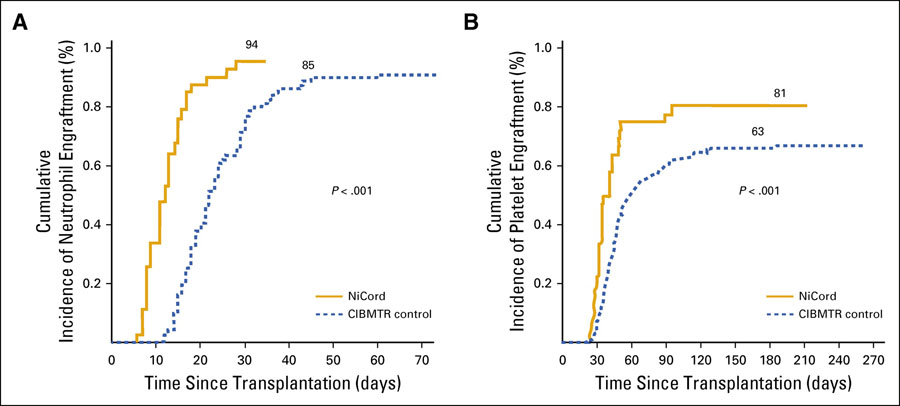
Shorter blood cell count recovery times may produce better outcomes for patients following transplant. Patients with compromised blood cell counts are more prone to infections. In these patients, even normally benign infections can pose a serious – even fatal – danger.
Compared to patients who received unexpanded UCB treatments, the incidence of Graft-Versus-Host disease (GVHD) with those who received the expanded UCB cells decreased at 100 days following transplant from 58% to 44%. GVHD is a serious and potentially fatal complication among transplant patients. Over time, the incidence of moderate to serious GVHD between transplant types was similar. Likewise, the 2-year disease-free survival rate among NiCord recipients (43%) was similar to unexpanded UCB recipients (45%). The overall two-year survival rates among both study groups were also similar.
The study demonstrated that the use of expanded cord blood on adult patients was both safe and effective. In addition, the use of expanded cord blood may potentially enable patients to recover faster and avoid some serious complications during the initial post-transplant phase.1
This study is an example of some of the exciting work that is taking place with umbilical cord blood. It also demonstrates that new techniques may increase the effectiveness and safety of UCB transplants. If you would like more information about banking your child’s umbilical cord blood for future stem cell treatments, please contact us at the New England Cord Blood Bank at (888) 700-2673 or visit the rest of our website at https://www.cordbloodbank.com.
- Horwitz M.E., Wease S., Blackwell B., Valcarcel D. et al. 2018. Phase I/II study of stem-cell transplantation using a single cord blood unit expanded ex vivo with nicotinamide. Journal of Clinical Oncology. DOI: 10.1200/JCO.18.00053